Organoids
Every Cell has a Story to Tell.
Let’s Discover Yours
Organoids are precious. They require expertise, months of effort, and significant resources to develop, making every sample valuable. Often patient-derived and biologically complex, each organoid provides a physiologically relevant model of human neurodevelopment or neurological disease. Their full value can only be realized with the right tools.
Our High-Density Microelectrode Array (HD-MEA) technology is designed to make every organoid count, enabling detailed functional characterization at single-cell and network levels, even at early stages of development, with unmatched spatial and temporal resolution, in both acute and longitudinal recording paradigms

Uncover the full functional profile of every organoid
Your organoids are precious, make them count and unlock insights that optical imaging alone cannot provide. Reveal how organoids fire, connect, and develop, even at early stages, with functional insights captured at subcellular, single-cell, and network level by our HD-MEA technology.
Greater reproducibility with fewer samples
Minimize biological variability with reproducible, high-quality functional data across wells and timepoints. Our high electrode count ensures reliable, statistically robust results, especially critical when organoid samples are limited.
Always at the right spot
Optimize recording quality with our flexible, high-density electrode configuration, ensureing you capture signals directly beneath active cells, including the center of the organoid. Obtain high-quality data across your organoids without requiring perfect placement every time.
Biologically relevant and non-invasive functional readouts
Capture the true activity of your organoids without compromising viability. Our system records from connected networks on the organoid surface, without invasive probes or dyes, providing biologically relevant signals that reflect how your model actually behaves.
Fit for a wide range of organoids
Organoids vary in protocol, structure, and handling, but our technology adapts. Whether you are performing acute or longitudinal recordings, our platform has been proven across a broad range of organoid use cases by researchers worldwide.

Functional characterization of organoids
Record diverse organoid models at high-resolution, both at single timepoints and longitudinally, thanks to the MaxOne Single-Well and MaxTwo Multi-Well HD-MEA Systems. With 26,400 electrodes per well, precisely capture network dynamics and single-cell activity across organoids spanning different protocols, complexities, and maturational stages.
Case studies
Functional profiling of different neural organoid models
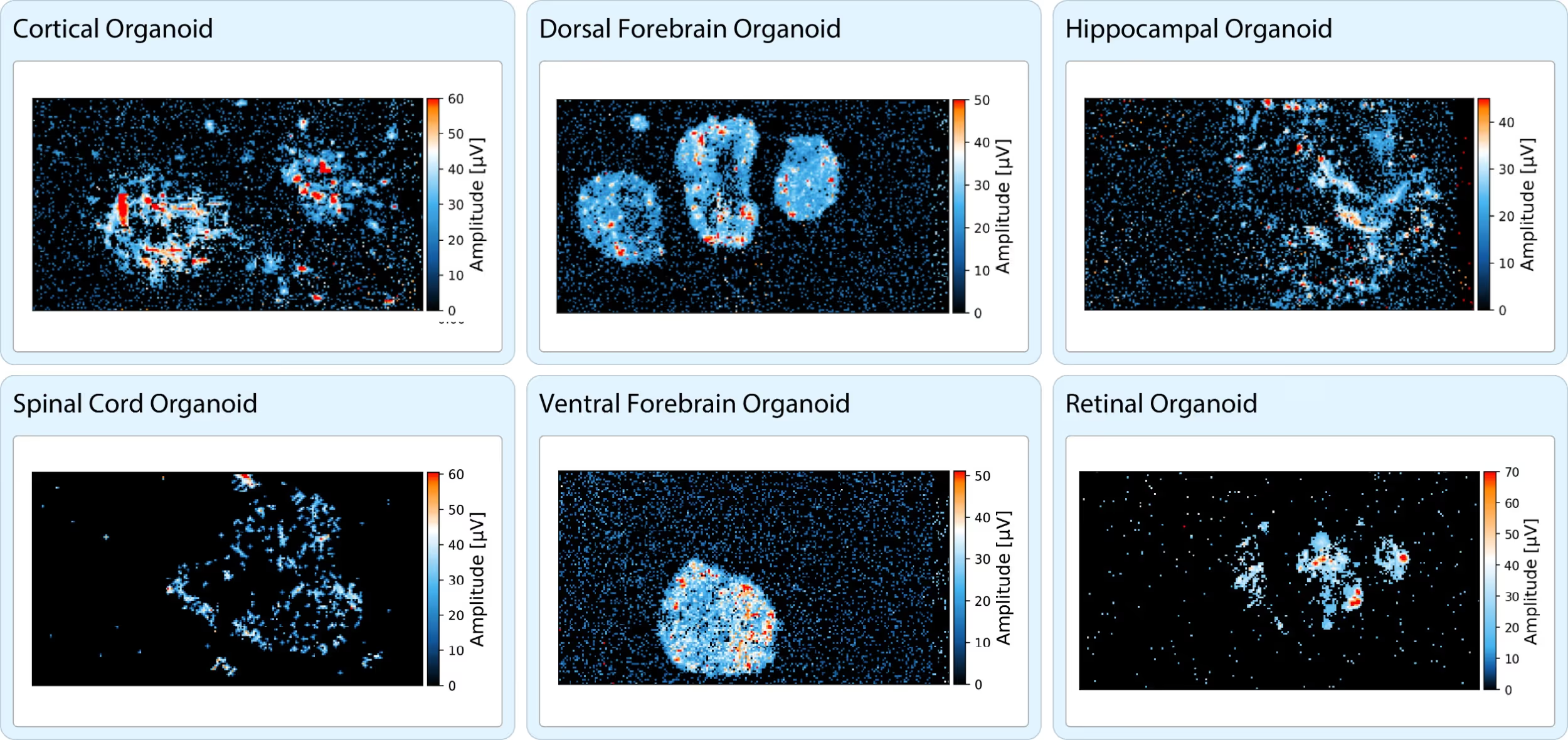
For every neural organoid model, extract a comprehensive functional signature using our HD-MEA technology. The representative recordings below highlight the high-quality characterization of a wide range of organoids, extending down to sub-cellular resolution.
Whole sample functional characterization of neural organoids.
ActivityScan Assay heatmaps depicting the activity patterns from representative neural organoids of various types including cortical, dorsal-forebrain, hippocampal, spinal cord, ventral-forebrain and retinal organoids.
Data courtesy of multiple MaxWell Biosystems’ users.
Acquire instant readouts from your organoids
Extract rich functional data acutely from organoids with ease using our HD-MEA technology. While multiple approaches exist to stabilize organoids on the MaxOne Chips and MaxTwo Multi-Well Plates, an innovative, user-driven patented approach involves a Liquid Holder known as perfluorodecalin (PFD) (Duenki et al., Advanced Healthcare Materials, 2025). This biocompatible liquid gently anchors organoids onto the electrode array, ensuring optimal contact without compromising tissue integrity. Perform reliable acute recordings of your organoids with ease.
Acute recording of a cortical organoid
Top: Zoom into the MaxOne Chip electrode array (left) showing a light microscopy image of the organoid on the chip (middle), with the corresponding recording overlaid, where each red triangle represents an action potential. To the right, the electrical footprint of a single neuron is mapped across multiple electrodes.
Bottom: Example functional readouts from MaxLab Live, including an ActivityScan Assay heatmap depicting the organoid-wide activity patterns (left) and Network Assay raster and network activity plots (middle, right) highlighting synchronous network bursts across numerous cells.
Data collected in collaboration with the Biomolecular and Cellular Engineering Lab headed by Professor Yoshiho Ikeuchi at the Institute of Industrial Science, University of Tokyo.
Track organoid development over time
Functionally characterize your organoids longitudinally in detail (for example, to monitor their maturation) with our HD-MEA technology. Obtain readouts to illustrate how functional phenotypes evolve over time, without disrupting or repositioning the sample.
Longitudinal recording of midbrain organoids co-cultured with microglia (assembloids).
Top: ActivityScan Assay heatmap of a representative assembloid showing steady increase in spike amplitude up to 28 days in vitro (DIV) from plating day. Network Assay raster and network plots revealed increasing synchrony and burst frequency from as early as two weeks post-plating.
Bottom: Zoomed view showing a light microscopy image of assembloid on a MaxOne Chip with the corresponding recording overlaid. Each red triangle represents an action potential. Bottom right, the electrical footprint of a single neuron recorded across multiple electrodes and the raw trace from a single electrode are displayed.
Data generated in-house using iPSC-derived midbrain organoids provided by OrganoTherapeutics.
Selected Resources
Preconfigured neuronal firing sequences in human brain organoids

A human striatal-midbrain assembloid model of alpha-synuclein propagation
Human neural organoid microphysiological systems show the building blocks necessary for basic learning and memory

FGF8-mediated gene regulation affects regional identity in human cerebral organoids

Functional neuronal circuitry and oscillatory dynamics in human brain organoids

Functional imaging of brain organoids using high-density microelectrode arrays

Acute Brain Organoid Plating Protocol with Liquid Holder
Acquire functional HD-MEA recordings from your brain organoids in no time with this unique and easy-to-use protocol.
Brain Organoid Supplementary Guide
Discover helpful tips for optimizing your organoid and spheroid cultures and their recordings on the MaxOne and MaxTwo systems.
MaxOne+ and MaxOne Brain Organoid Plating Protocol
Use this brain organoid plating protocol for MaxOne+& MaxOne to achieve optimal attachment and enable longitudinal electrophysiology recordings of your neural organoids.
MaxTwo Brain Organoid Plating Protocol
Use this Brain Organoid Plating Protocol for MaxTwo to achieve high-throughput, longitudinal electrophysiology recordings of your neural organoids.
Sliced Brain Organoid Protocol
This protocol describes the preparation, slicing, and recovery of human brain organoid sections optimized for high-density electrophysiological recordings on MaxWell Biosystems platforms
Organoids Application Brochure
MxW - ACROBiosystems Application Note
Acute recordings of spinal cord organoids generated via a commercially available kit from ACROBiosystems, performed on MaxTwo 6-Well Plate with Liquid Holder.
Breathe-Easy Foil MaxTwo 6-Well Plate Video Instruction
See how easy it is to apply the Breathe-Easy® sealing membranes on our MaxTwo Multiwell Plates, minimizing evaporation and ensuring optimal conditions for your sample.
MaxOne Organoid Plating Video Instruction
Plating organoids on MaxOne Chips has never been so easy, thanks to our detailed step-by-step instructions, with practical tips at key stages.
MaxTwo Brain Organoid Plating Protocol Video Instructions
Plating organoids on MaxTwo Multiwell Plates has never been so easy, thanks to our detailed step-by-step instructions, with practical tips at key stages.