MaxLab Live
AxonTracking Assay
A Novel Approach
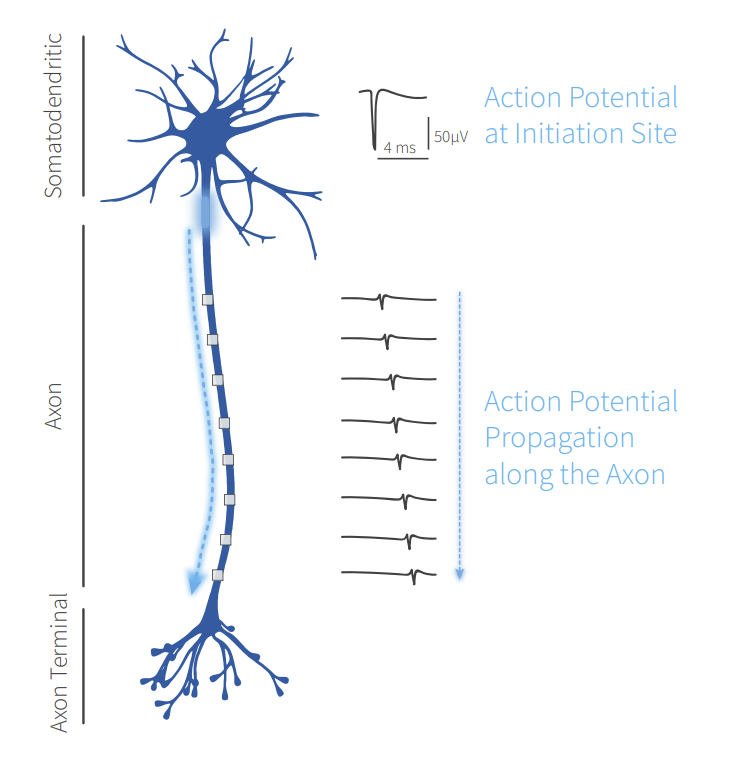
Neurons communicate within a network via action potentials (APs) propagating along axons. The capability to access axonal physiology is crucial for studying information processing among neurons in healthy and diseased states. However, axonal signals are difficult to measure at a large-scale. Therefore, the combination of reliability, ease of use, throughput, long-term and non-invasive measurement are necessary to monitor and understand neuronal function at a scale that was previously not possible.
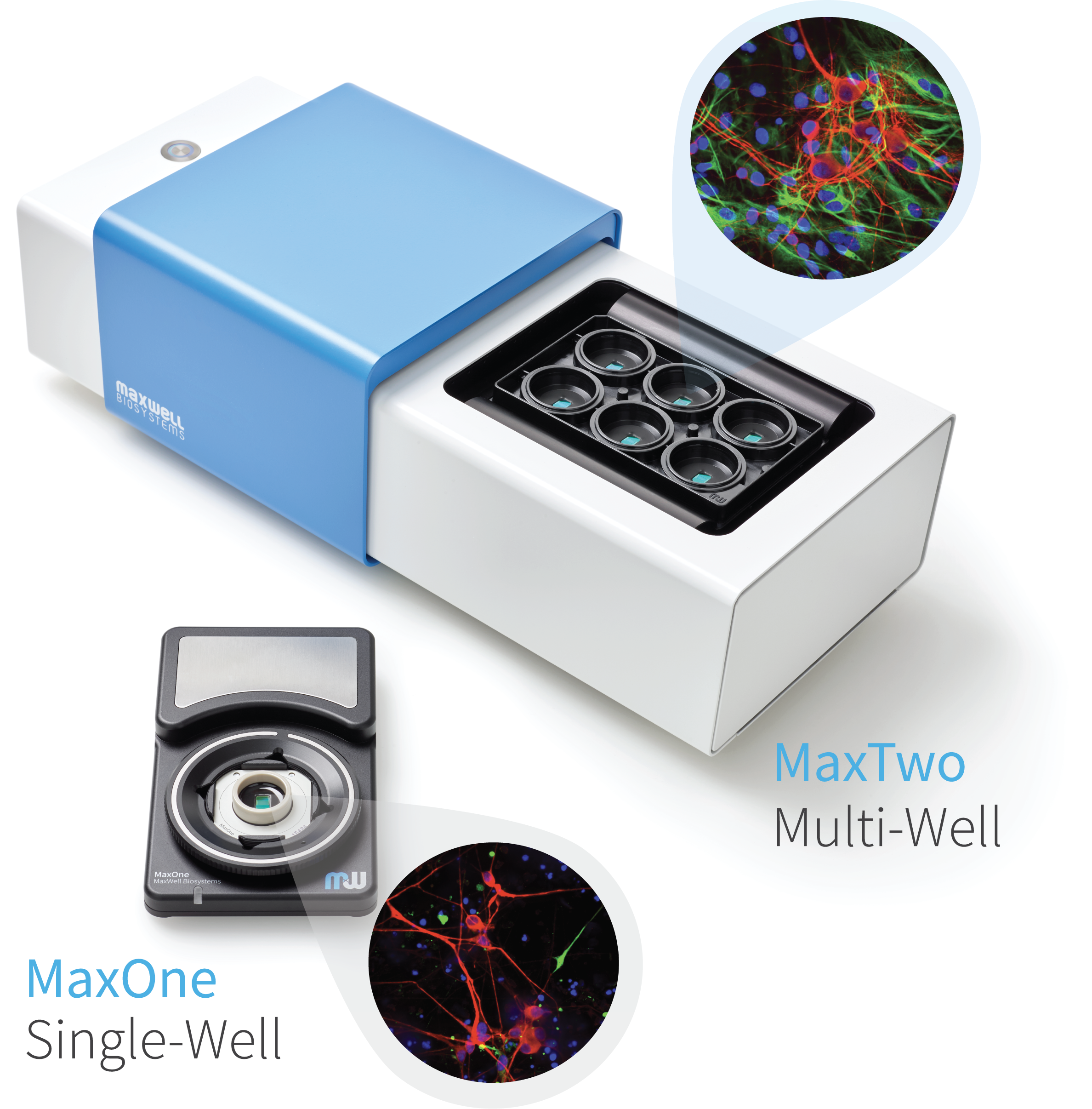
High-density microelectrode array (HD-MEA) measurements at unprecedented resolution and high signal quality, using MaxOne and MaxTwo systems, allow to detect the AP propagation from the initiation site down to distal axonal branches. With the AxonTracking Assay, the identification of the axonal paths is fully automated at the micrometer scale. This live-cell recording and analysis provides novel functional and structural readouts applicable for phenotypic characterization, disease modeling, and drug screening studies.

Automated | Long-Term | Label-Free |
|---|---|---|
| The fully automated platform is easy to use and allows for simultaneous recordings of multiple neurons and axonal branches in multiple wells. | Characterize neuronal maturation, development or treatment effects by recording from your culture over multiple days and weeks. | The electrical recordings are noninvasive and label-free, which avoids introducing side effects from dyes etc. |
HD-MEA Technology for Recording Axonal Signals
Powered by MaxOne and MaxTwo

Key advantages:
| High Spatio-Temporal Resolution Reconstruct axonal paths by tracking Action Potential propagation at thousands of sites, thanks to the densely packed microelectrode array. | |
| Large Sensor Area Detect long axonal branches of multiple neurons at the same time with a large sensor area, applicable for 2D and 3D samples. | |
| High Signal Quality Catch the smallest signals propagating along axons, down to single micro-volts-range, with low-noise recording channels |
Experimental Workflow
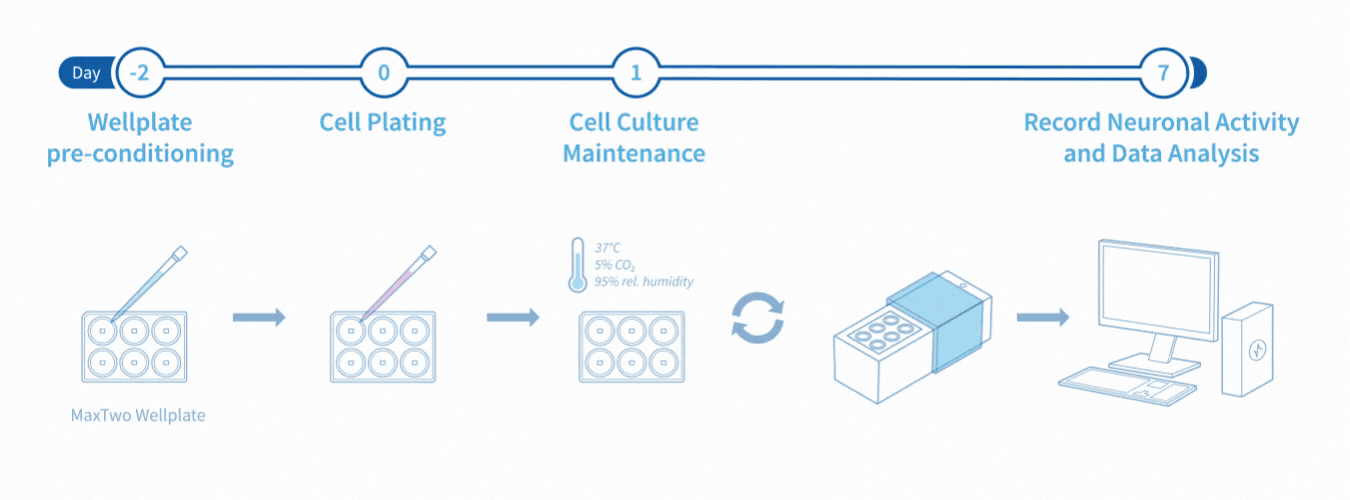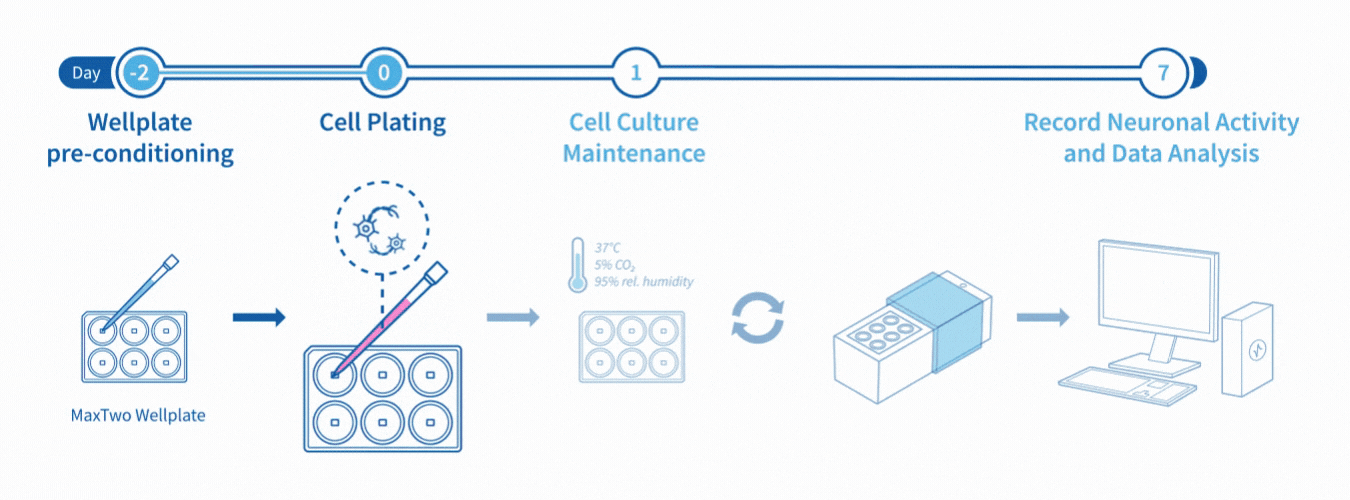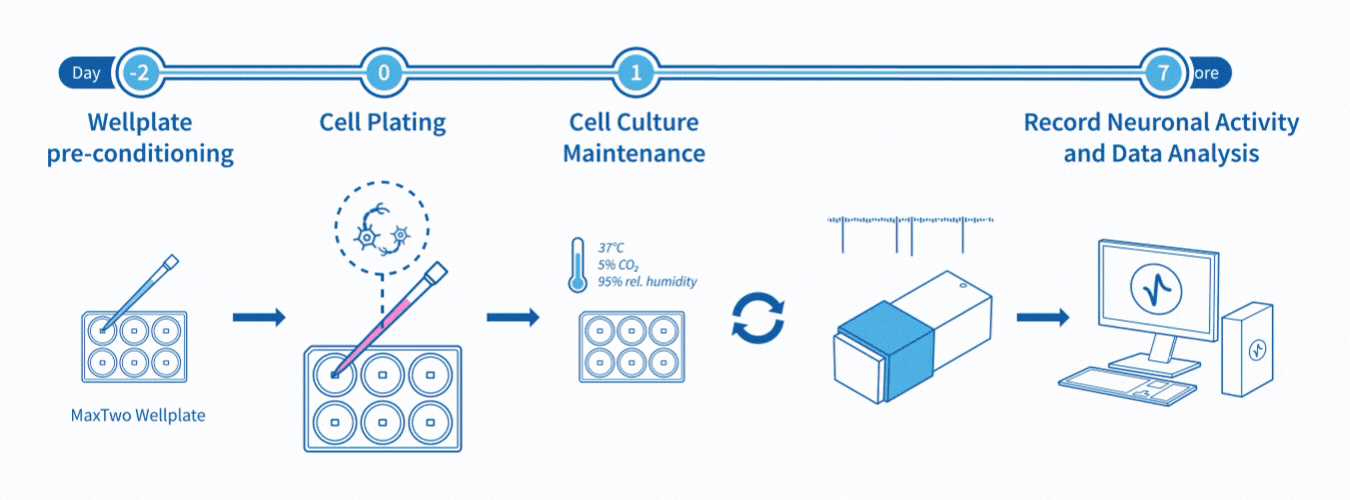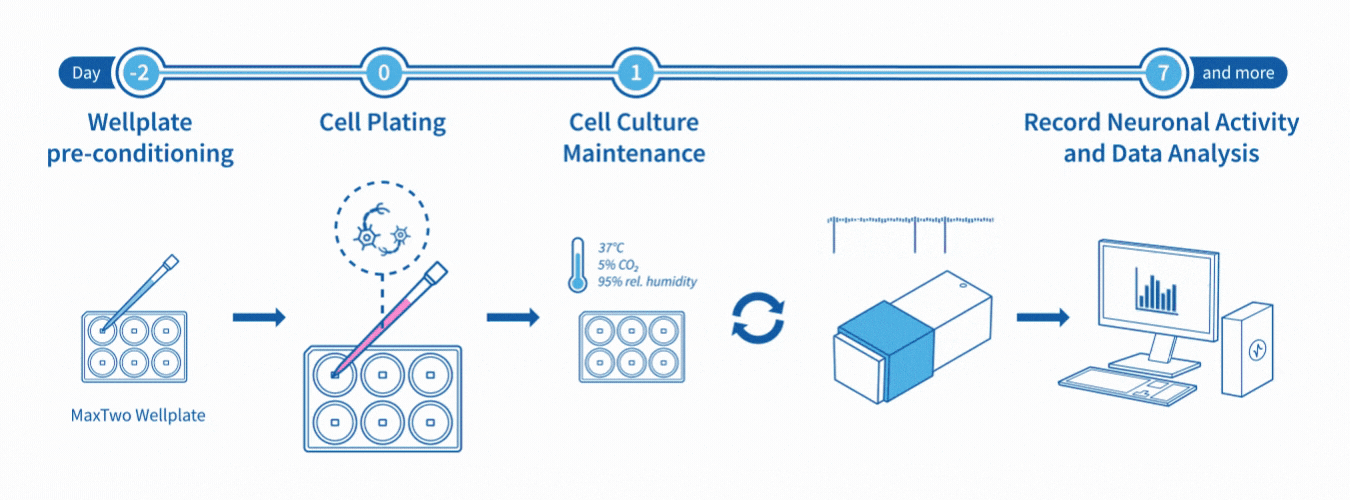

Assay Workflow
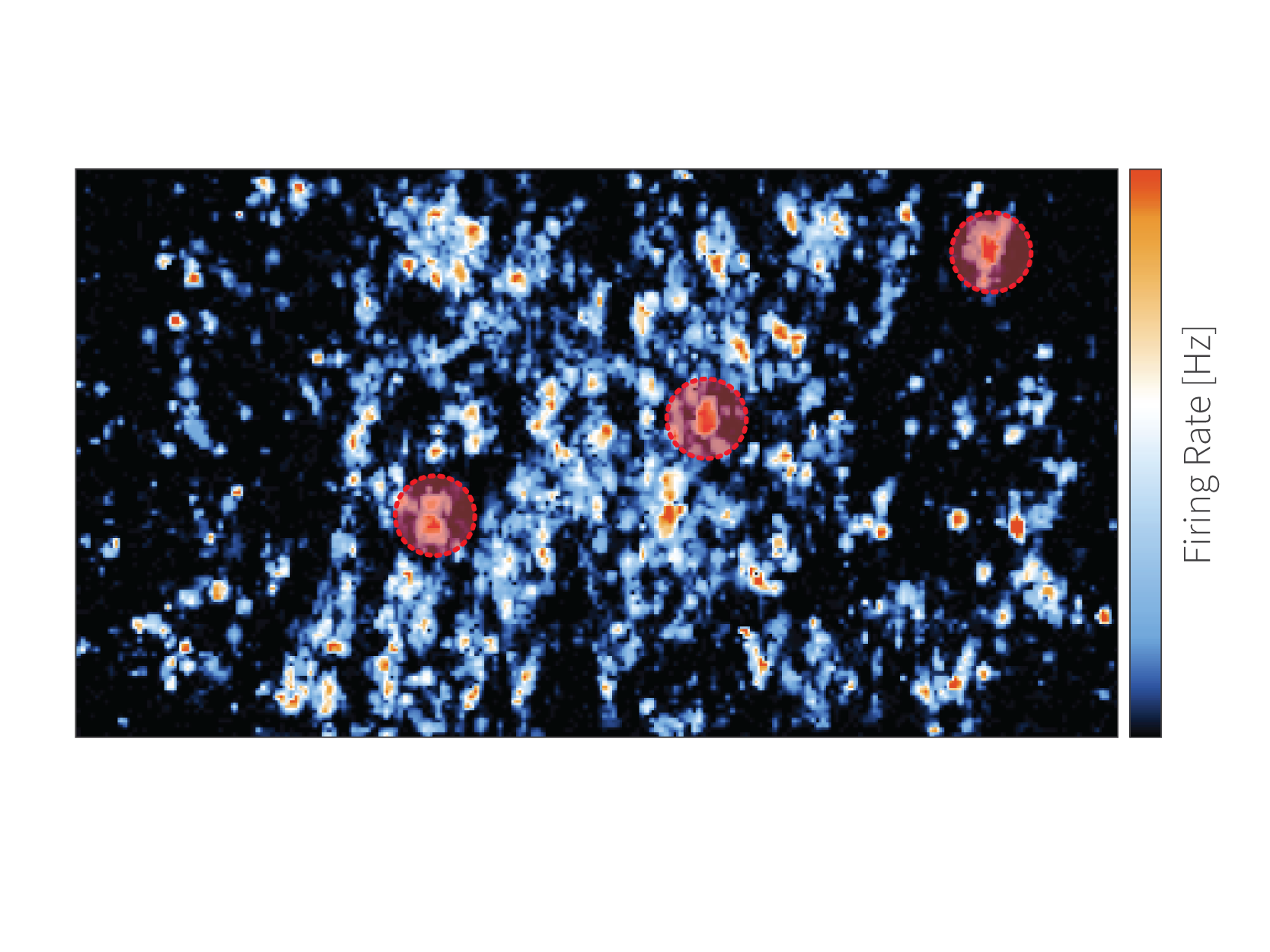  | RecordRecord the active neurons identified with the ActivityScan Assay. |
  | ProcessReveal the axonal morphologies through a series of processing steps: ◉ Spike sorting ◉ Spike-triggered averaging ◉ Footprint extraction |
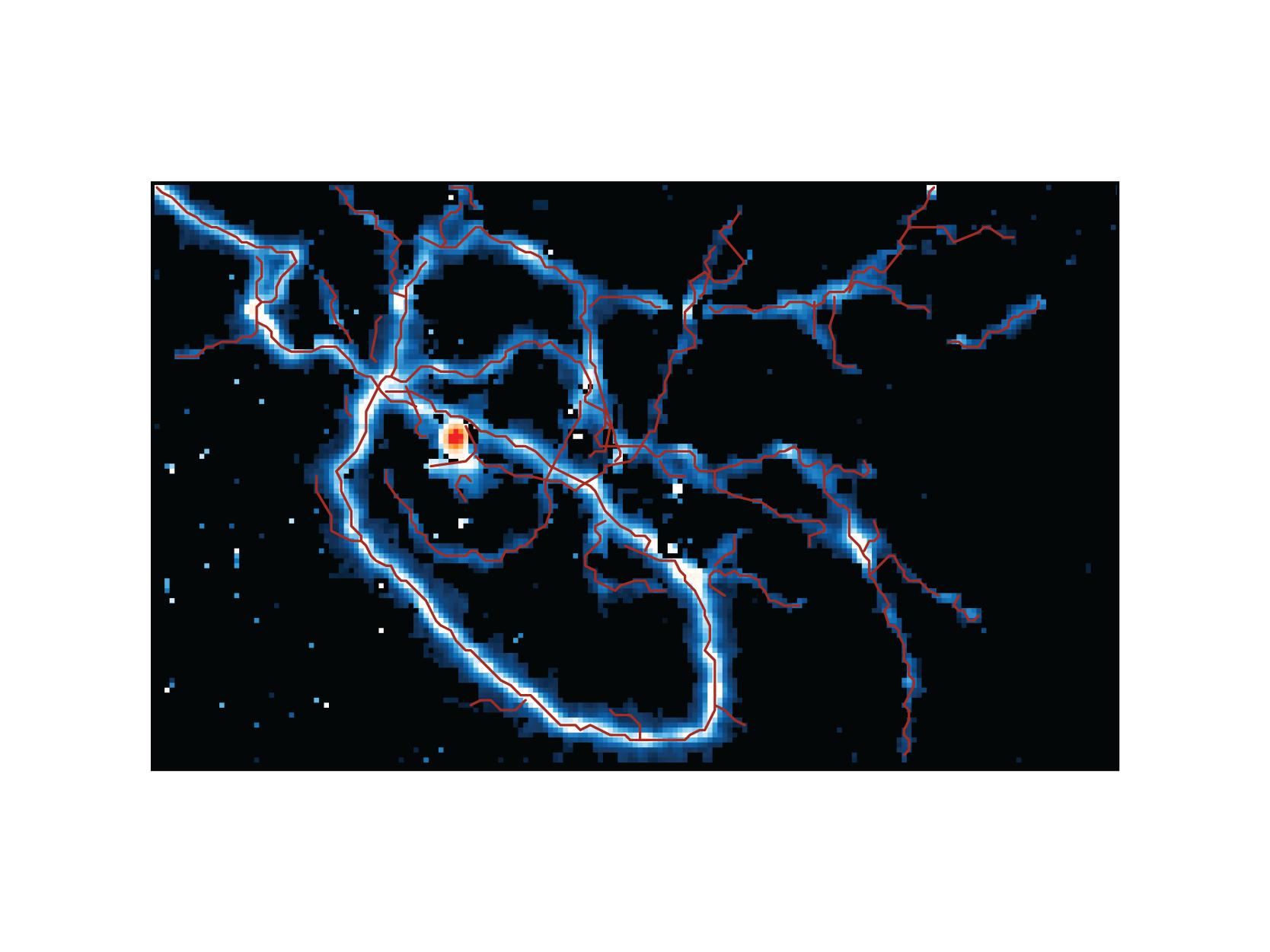  | AnalyzeIdentify individual axonal branches and reconstruct the morphology of the neurite outgrowth using an unsuper- vised object-tracking algorithm. |
Metrics
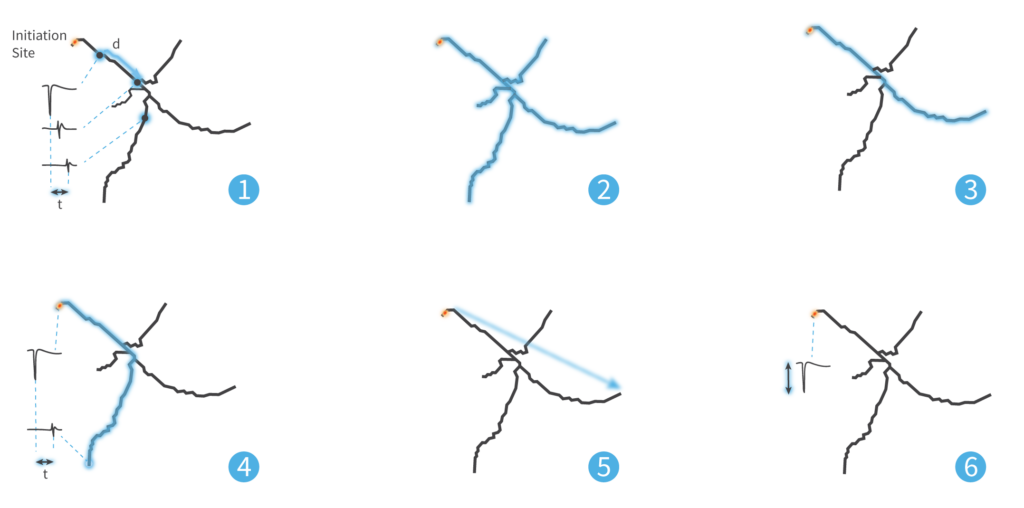

Legend
1 – Neuron Conduction Velocity
2 – Total Detected Axon Length
3 – Longest Branch Length
4 – Longest Latency
5 – Longest Distance from Initiation Site
6 – Amplitude at Initiation Site
Results
AxonTracking Assay in Human Neurons
Propagating APs along axonal processes recorded from different human induced pluripotent stem cells-derived (iPSC-derived) neuronal cell lines.
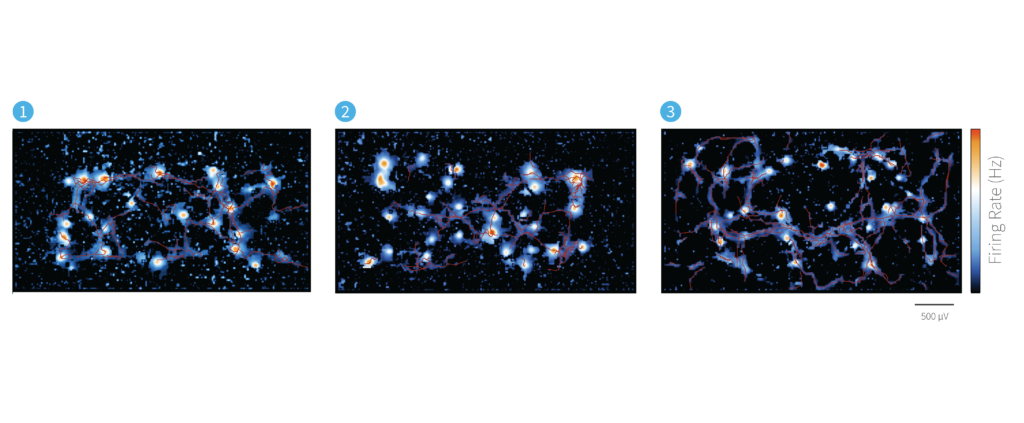

Legend
1 – Human Glutamatergic Neurons
2 – Human Dopaminergic Neurons
3 – Human Motor Neurons
AxonTracking Assay in Long-Term Neuronal Cultures
Neurons and propagating APs can be resolved even in cultures with long and dense axonal processes (iPSC derived glutamatergic neurons, DIV 63)
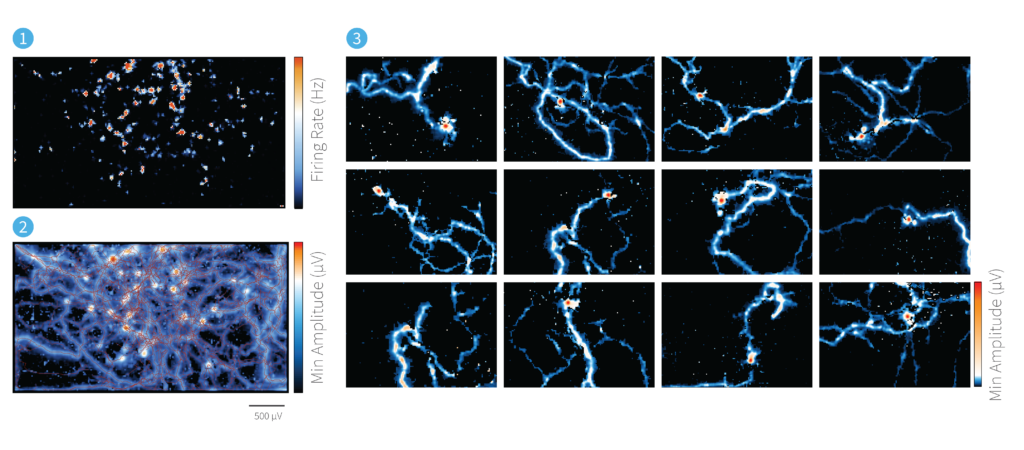

Legend
1 – Activity map
2 – AxonTracking Assay
3 – Propagating AP of individual neurons resolved
AP Propagation Along Axons
Propagating APs along the axonal processes of a rat primary cortical neuron shown in a time-series:


References
Bakkum, D. J., Frey, U., Radivojevic, M., Russell, T. L., Müller, J., Fiscella, M., Takahashi, H., & Hierlemann, A. “Tracking axonal action potential propagation on a high-density microelectrode array across hundreds of sites.” Nat Commun. 4, 2181 (2013).
Bullmann, T., Radivojevic, M., Huber, S. T., Deligkaris, K., Hierlemann, A., & Frey, U. “Large Scale Mapping of Axonal Arbors Using High-Density Microelectrode Arrays.” Front. Cellular Neurosci. 13, 404 (2019).
AxonTracking Assay Brochure
Want to try this with your cells?
Support
We provide training and support on all aspects of MaxLab Live Software assays and modules. To learn more about these MaxLab Live Software Modules, please Contact Us.
Discover More
  |   |
 English
English


