Scale-up your experiments
Discover the full potential of HD-MEAs at throughput: scalable, precise, and designed for automation.
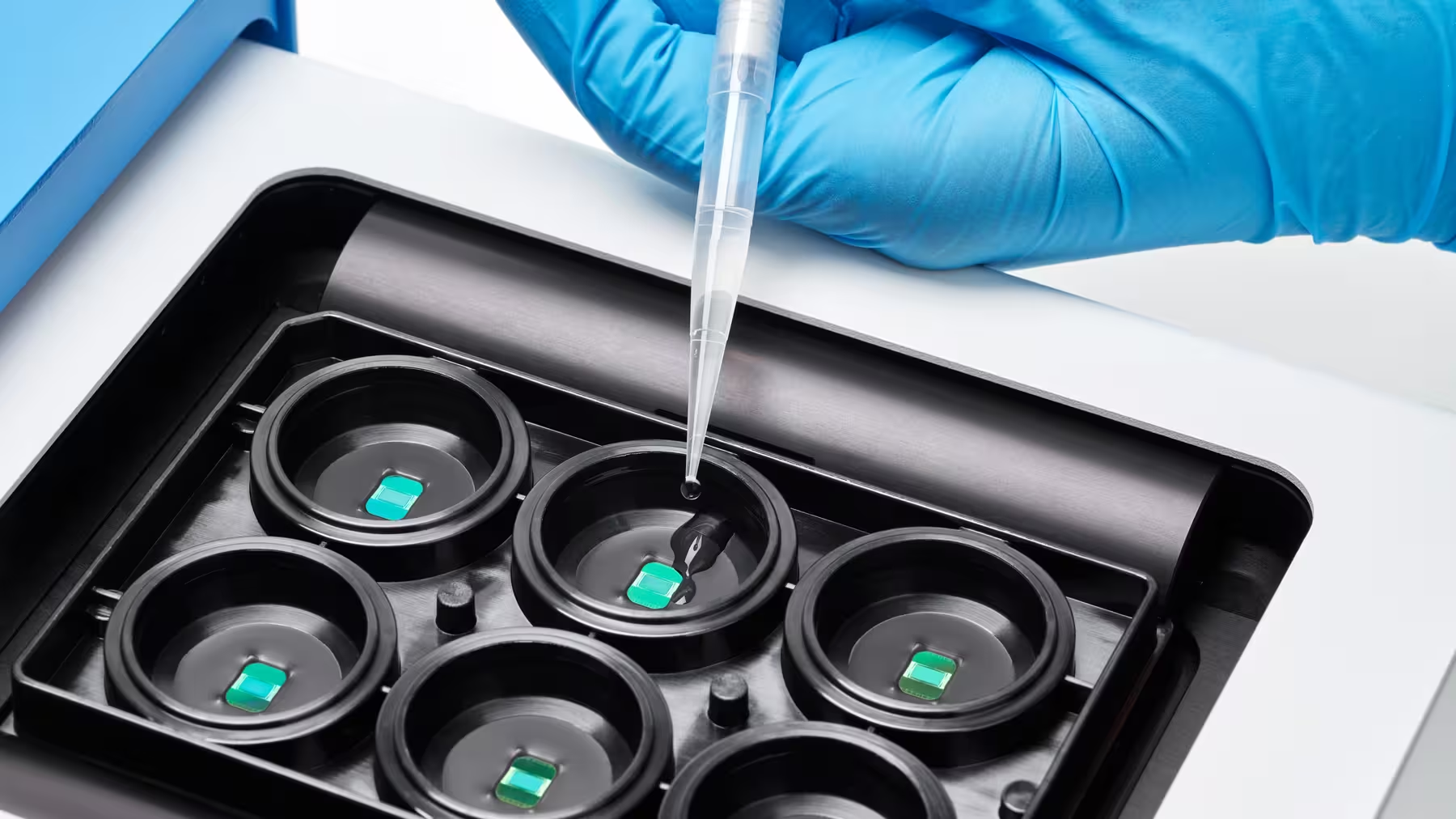
Consistent high resolution in every well
Increase throughput without compromising data quality. Each well features 26,400 electrodes at a density of 3,265 electrodes/mm², maintaining single-cell and subcellular resolution across the entire plate.
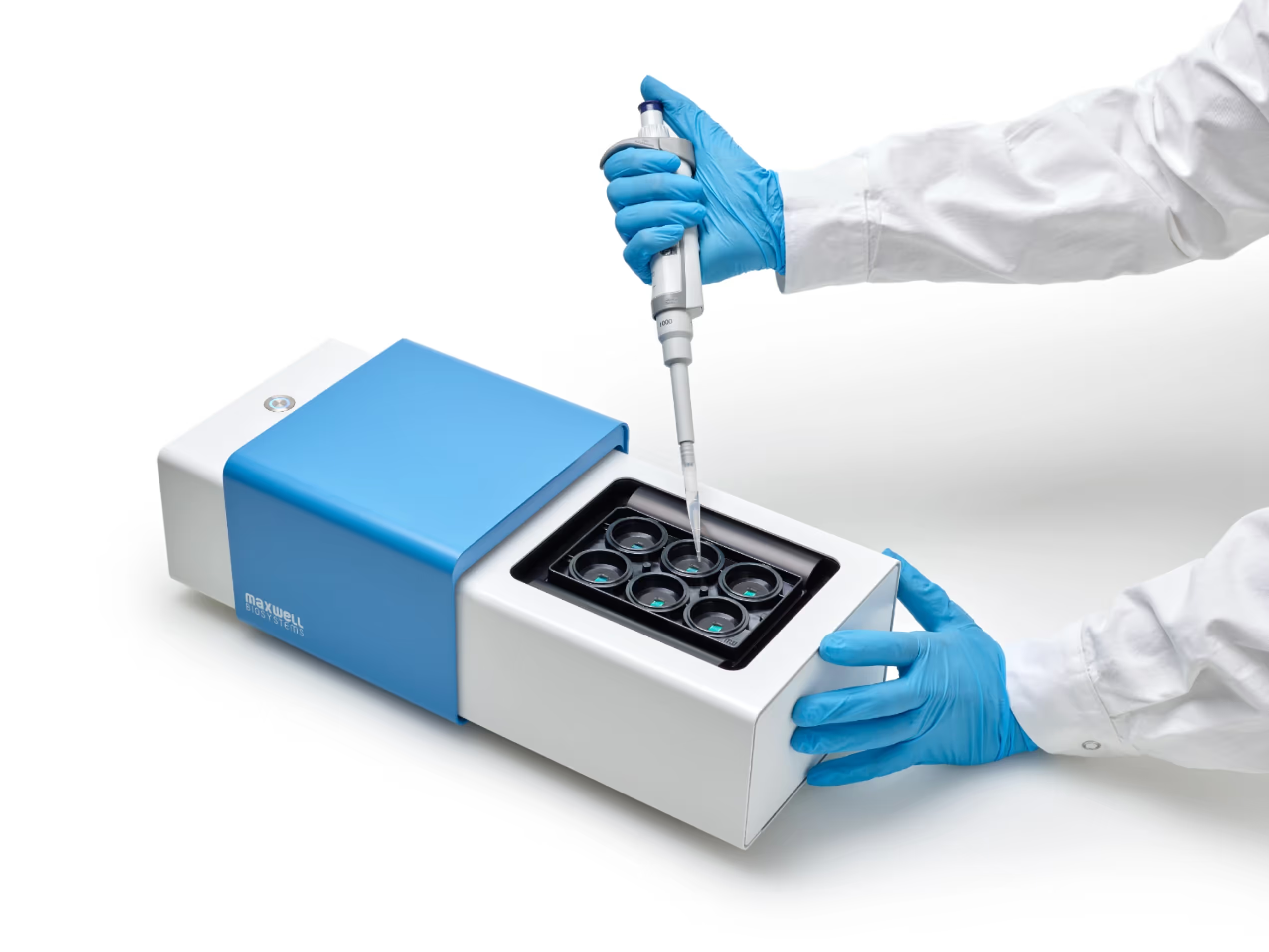
Automation-tested platform
Engineered for compatibility with standard laboratory automation tools. MaxTwo is designed for seamless integration with liquid handlers, plate movers, and robotic systems, enabling plug-and-play operation for automated workflows.
Efficient large-scale data handling
Manage high volumes of recordings with ease. Sample annotation, batch processing and report generation streamline analysis across multiple wells and experiments, accelerating insight and decision-making.
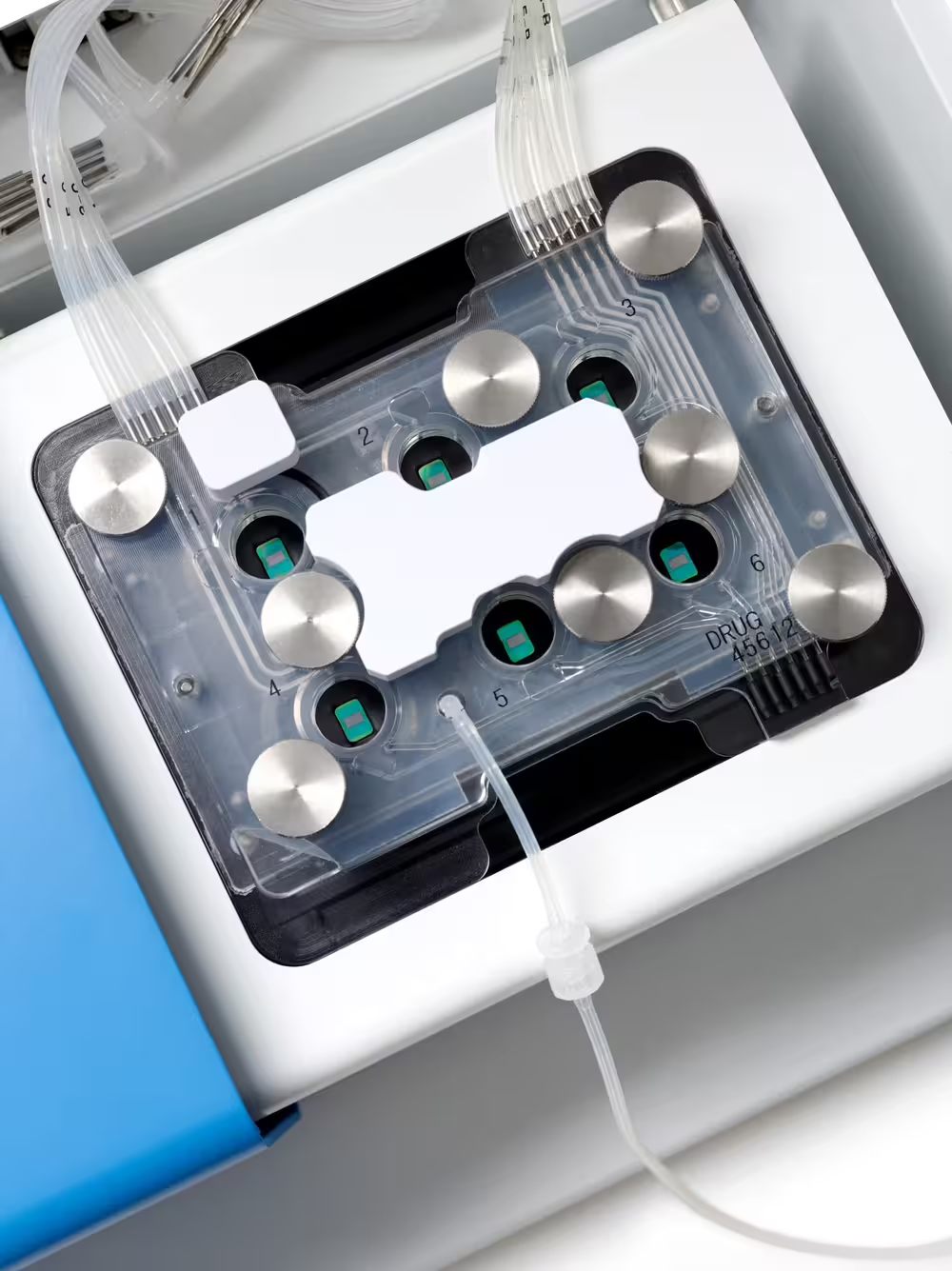
Fully integrated perfusion system
Control experimental conditions across all wells with the first fully integrated perfusion system in a MEA multi-well platform. Maintain sample viability with minimal noise while recording from all wells in parallel.






.avif)



.avif)
.avif)